Our Senior Research and Development Scientist, Yvonne Helbert, recently spoke with Alex Beadle from Analytical Cannabis about why the PathoSEEK Microbial Safety Testing Platform is the best microbial testing method for cannabis. They discussed the complexity of the cannabis matrix, the advantages of qPCR, the pitfalls of plating, and the importance of validation. Here are some of the key takeaways. You can read the full interview at Analytical Cannabis.
Sticky Cannabis Flower Makes Sampling Tricky
“The flower is lipid-rich, making it very sticky. This presents a challenge because lipid-rich matrices cannot be sampled with simple aqueous emersions. The lipids need to be dissolved in water to fully sample such a matrix which requires a strong lysis buffer. While quick and simple aqueous preps are appealing for quick labor-free extraction they fail to fully sample the microbial diversity of the flower.
“Medicinal Genomics has developed and validated the SenSATIVAx DNA extraction kit and PathoSEEK qPCR detection assays specifically for cannabis microbial testing. This is important because many other microbial testing solutions for cannabis have been lifted from the food industry and as far as we know have not been validated on cannabis.” Download our Validation Document.
DNA-Based Methods Can Detect Harmful Endophytes
“In addition to the many microbes that live on the plant’s surface, there are also endophytes, which are bacteria or fungus that live inside the plant without causing apparent disease to the plant. Since most culture-based techniques rely on viable cells growing in a medium for detection, they do not lyse plant cells open to see the endogenous pathogens like DNA-based methods do.
“Aspergillus is a known plant endophyte, and four Aspergillus species (flavus, fumigatus, nigerand terreus) are known human pathogens. It is therefore imperative that the flower sample be lysed so that the endophytes are accurately captured.”
qPCR is Active. Plating is Passive
“qPCR detection assays actively look for specific DNA sequences shared by a family of microbes. If the DNA is present, qPCR will detect it. This active approach means we can design species-specific assays that target only pathogenic species, such as Aspergillus and STEC E.coli.
“Culture plating is much more passive. You are essentially placing cells in a growth medium and hoping it’s favorable enough for them to grow so you can count them. The problem with that is only a fraction of microbes culture. And if you have multiple microbes in the same medium, they will compete with one another. We have demonstrated that the makeup of the microbial population after culturing can be radically different than what is actually on the plant.” Read our peer-reviewed study that demonstrates this
qPCR is a Widely Used Method
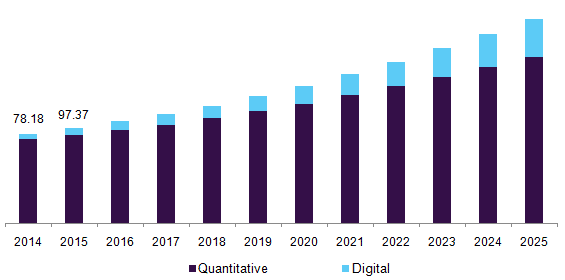
“qPCR techniques are widely used in gene expression, genotyping, pathogen detection, viral quantification, prenatal genetic testing, blood screening, oncology testing and much more. The qPCR market is considered to be the “gold standard” for accurate, sensitive and fast quantification of nucleic acid sequences. The global qPCR and digital PCR (dPCR) market is expected to reach USD 6.3 billion by 2025, according to a report by Grand View Research, Inc.”